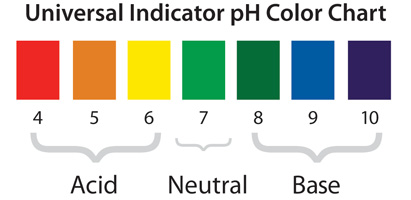
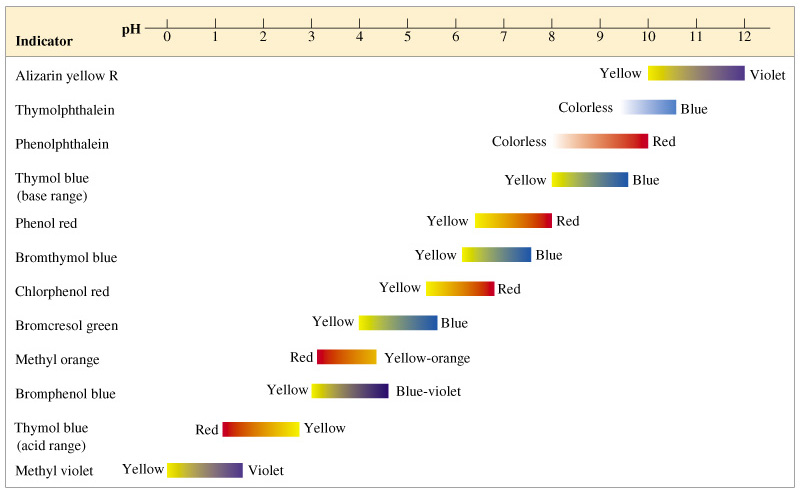
What is a base?
Something considered a base is something that can donate a hydroxide ion. It also is something that accepts Hydrogen ions.
Something considered a base is something that can donate a hydroxide ion. It also is something that accepts Hydrogen ions.
Examples of basic:Sodium Hydroxide - NaOH
Potassium Hydroxide - KOH
Ammonium Hydroxide - NH4OH
Calcium Hydroxide - Ca(OH)2
Magnesium Hydroxide - Mg(OH)2
Barium Hydroxide - Ba(OH)2
Aluminum Hydroxide - Al(OH)3
Ferrous Hydroxide or Iron (II) Hydroxide - Fe(OH)2
Ferric Hydroxide or Iron (III) Hydroxide - Fe(OH)3
Zinc Hydroxide - Zn(OH)2
Lithium Hydroxide - LiOH
Ammonia - NH3
Sodium Hydrogen Carbonate - NaHCO3
Sodium carbonate - Na3CO3
sodium hydride - NaH
Characteristics:
Potassium Hydroxide - KOH
Ammonium Hydroxide - NH4OH
Calcium Hydroxide - Ca(OH)2
Magnesium Hydroxide - Mg(OH)2
Barium Hydroxide - Ba(OH)2
Aluminum Hydroxide - Al(OH)3
Ferrous Hydroxide or Iron (II) Hydroxide - Fe(OH)2
Ferric Hydroxide or Iron (III) Hydroxide - Fe(OH)3
Zinc Hydroxide - Zn(OH)2
Lithium Hydroxide - LiOH
Ammonia - NH3
Sodium Hydrogen Carbonate - NaHCO3
Sodium carbonate - Na3CO3
sodium hydride - NaH
Characteristics:
- Slippery
- Bitter
- Conduct Electricity
- Don't react with metals
- turn red litmus paper blue