Definition:
The Limited amount of solute that can dissolve in a specific amount of solvent.
Saturated and Unsaturated:
When a solution is saturated is when you have added too much solute that no more can dissolve.
When a solution is unsaturated is when you continue to dissolve more solute.
The Limited amount of solute that can dissolve in a specific amount of solvent.
Saturated and Unsaturated:
When a solution is saturated is when you have added too much solute that no more can dissolve.
When a solution is unsaturated is when you continue to dissolve more solute.
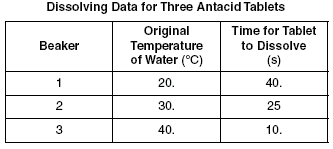
Solubility Rate: how fast a solute can dissolve in a solvent.
The three factors that affect solubility rate:
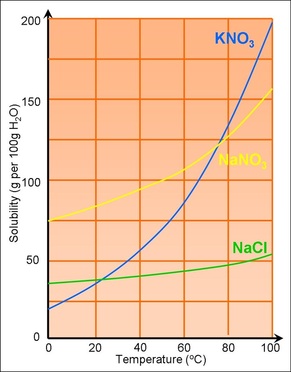
Temperature: Because if the solute changes in temperature the solubility may decrease.
Two examples of temperature are:
Temperature: Because if the solute changes in temperature the solubility may decrease.
Two examples of temperature are:
- Endothermic reactions: Increase in temperature
- Exothermic reactions: Decrease in temperature
Solute or solvent:
If there is a polar solute it must dissolve in a polar solvent.
Pressure: Pressure increases the solubility of a solvent.
If there is a polar solute it must dissolve in a polar solvent.
Pressure: Pressure increases the solubility of a solvent.
Explanations of Solute and Solvent:
If a solute in dissolved in water then it becomes a solvent.
A solvent can be a liquid, solid, or gas that is mixed with another solvent to form a solution.
A solution is a mixture, usually a homogeneous mixture.
If a solute in dissolved in water then it becomes a solvent.
A solvent can be a liquid, solid, or gas that is mixed with another solvent to form a solution.
A solution is a mixture, usually a homogeneous mixture.